Ipca Laboratories Walk-In Interview June 2025 – Career Opportunities at Silvassa (Piparia) OSD Unit
Are you a passionate and experienced pharmaceutical or engineering professional seeking rewarding career opportunities in one of India’s most respected pharmaceutical companies? Ipca Laboratories Limited invites qualified and dynamic candidates for a Walk-In Interview on 29th June 2025 (Sunday) for multiple positions at their state-of-the-art Formulation (OSD) Unit in Piparia, Silvassa.
This walk-in drive offers a golden chance for professionals across various departments like Production, Quality Assurance, Quality Control, Engineering, EHS, HR, and IT to become part of a globally trusted pharmaceutical brand. Here’s everything you need to know to secure your dream job at Ipca.
📍 Walk-In Interview Details
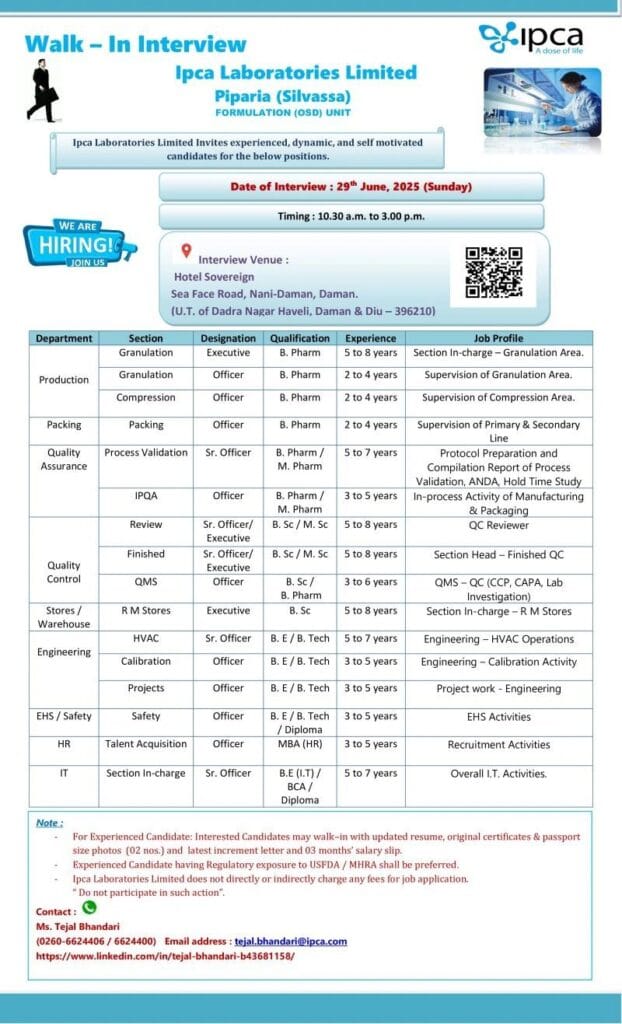
Date: 29th June 2025 (Sunday)
Time: 10:30 a.m. to 3:00 p.m.
Venue:
Hotel Sovereign
Sea Face Road, Nani-Daman, Daman
(U.T. of Dadra Nagar Haveli, Daman & Diu – 396210)
🎯 Departments & Vacancies
1. Production Department
Granulation & Compression (Formulation OSD)
| Designation | Qualification | Experience | Job Profile |
|---|---|---|---|
| Executive – Granulation | B. Pharm | 5 to 8 years | Granulation Area – Section In-Charge |
| Officer – Granulation | B. Pharm | 2 to 4 years | Supervision of Granulation Process |
| Officer – Compression | B. Pharm | 2 to 4 years | Supervision of Compression Activities |
2. Packing Department
| Designation | Qualification | Experience | Job Profile |
|---|---|---|---|
| Officer | B. Pharm | 2 to 4 years | Supervision of Primary & Secondary Packing Lines |
🛡️ Quality Assurance (QA) Department
| Section | Designation | Qualification | Experience | Job Profile |
|---|---|---|---|---|
| Process Validation | Sr. Officer | B. Pharm / M. Pharm | 5 to 7 years | Protocol & Report Compilation, ANDA, Hold Time Study |
| IPQA | Officer | B. Pharm / M. Pharm | 3 to 5 years | In-Process Activities during Manufacturing |
| Review | Sr. Officer / Executive | B. Sc / M. Sc | 5 to 8 years | Documentation & QA Review |
🧪 Quality Control (QC) Department
| Section | Designation | Qualification | Experience | Job Profile |
|---|---|---|---|---|
| Finished | Sr. Officer / Executive | B. Sc / M. Sc | 5 to 8 years | Section Head – Finished Product QC |
| QMS | Sr. Officer / Officer | B. Sc / B. Pharm | 3 to 6 years | QMS Activities – CCP, CAPA, Lab Investigation |
🏬 Stores/Warehouse
| Designation | Qualification | Experience | Job Profile |
|---|---|---|---|
| Executive – RM Stores | B. Sc | 5 to 8 years | Section In-Charge – Raw Material Stores |
⚙️ Engineering Department
| Section | Designation | Qualification | Experience | Job Profile |
|---|---|---|---|---|
| HVAC | Sr. Officer | B. E / B. Tech | 5 to 7 years | HVAC System Operation |
| Calibration | Officer | B. E / B. Tech | 3 to 5 years | Calibration Activities |
| Projects | Officer | B. E / B. Tech | 3 to 5 years | Project Management in Engineering |
🧯 EHS (Environment, Health & Safety)
| Designation | Qualification | Experience | Job Profile |
|---|---|---|---|
| Officer – Safety | B. E / B. Tech / Diploma | 3 to 5 years | EHS Implementation and Safety Compliance |
💼 Human Resources (HR)
| Designation | Qualification | Experience | Job Profile |
|---|---|---|---|
| Officer – Talent Acquisition | MBA (HR) | 3 to 5 years | Recruitment and Talent Management |
💻 Information Technology (IT)
| Designation | Qualification | Experience | Job Profile |
|---|---|---|---|
| Sr. Officer – IT | B.E (I.T) / BCA / Diploma | 5 to 7 years | Managing Overall IT Operations |
🔍 Key Instructions for Candidates
Carry the following documents:
Updated Resume
Two Passport-size Photographs
Original Certificates & Photocopies
Latest Increment Letter
Last 3 Months’ Salary Slips
Preferred: Candidates with regulatory exposure to USFDA / MHRA will be prioritized.
Note: Ipca Laboratories does not charge any fees for job applications or interviews. Beware of fraudulent job offers.
📞 Contact Information
For queries or details:
Contact Person:
Ms. Tejal Bhandari
📞 0260-6624406 / 6624400
📧 Email: [email protected]
🔗 LinkedIn Profile
💡 Why Choose Ipca Laboratories Limited?
Global Presence and Regulatory Recognition
Ipca is a well-known name in the global pharmaceutical industry, with approvals from USFDA, WHO-GMP, MHRA, TGA, and other major regulatory agencies.
State-of-the-Art Manufacturing Facilities
The Piparia (Silvassa) Formulation OSD Unit is a cutting-edge facility equipped with world-class technology, automation, and compliant with global manufacturing standards.
Work Culture that Empowers Growth
Ipca offers a professional work environment with a focus on employee development, continuous training, and internal promotions.
Attractive Compensation and Benefits
Competitive salary packages, performance-linked incentives, and numerous employee welfare initiatives make Ipca a preferred employer in the pharmaceutical sector.
🔚 Conclusion: Don’t Miss This Golden Career Opportunity
Whether you are an experienced pharmaceutical production officer, a quality control specialist, or a technical professional in engineering or IT, this walk-in interview by Ipca Laboratories Limited could be your next big career breakthrough.
Make sure to mark your calendar for 29th June 2025, prepare your documents, and walk into the venue with confidence. This opportunity may be the turning point that propels your career to new heights.
To Join In Whatsapp group Click below Link
https://chat.whatsapp.com/ERzsuXoDDnOLBZ3spkddn5
ITI JOB ALTERS WHATSAPP GROUP
https://chat.whatsapp.com/Kv3A7ymWIU7D1pZZaUrEiS
To Join Telegram Channel Click Below Link
https://t.me/careerpathwayjobs
To know the Latest Job updates click below Link
Career Pathway – Guide to your success

